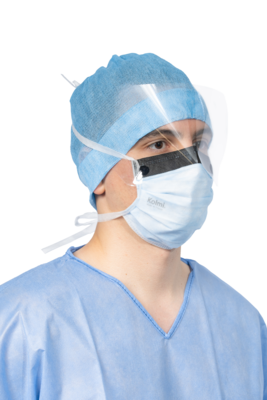
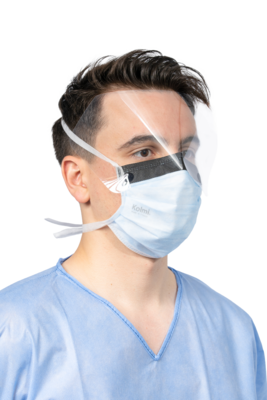
Medical mask Type IIR Op Air® One

Medical device : Class I - Type of medical mask : Type IIR - Tie-on - Foam strip + Anti-glare strip + Visor
Ref. M34101MNV-30
-
Dental -
Medical -
Industry -
Hygiene
FEATURES AND BENEFITS
TECHNICAL SPECIFICATIONS
PRODUCT INFORMATION
CERTIFICATIONS & STANDARDS
DOCUMENTS
Features and benefits
- Protects the environment from droplets emitted by the wearer while providing protection from splashes of biological fluids.
- Effective against bacteria, particles and viruses (BFE, PFE, & VFE)
- Kolmi masks feature the exclusive Origami folding for greater comfort and a perfect fit on the wearer's face.
- For maximum comfort, the inner layer is designed with a material that limits irritation to the face.
- With Feeldry technology the inner layer has been specially designed to absorb perspiration and avoid the feeling of humidity inside the mask.
- Clear polyester visor with anti-fog treatment on both sides.
- Foam strip improves comfort for the user and reduces fogging of the glasses.
- Anti-glare strip reduces the risk of glare in operating theatres.
- Hypoallergenic.
- Use by medical staff for different types of care and other people to reduce the risk of spreading infections, especially in epidemic or pandemic situations.
- Use by operating room staff in surgeries where there is a risk of splashing of biological fluids.
- Use by medical staff wearing glasses or using binocular magnifiers.
- Use by medical staff in operating theatres or in areas of high light intensity.
- Recommended for sensitive skin.
Technical specifications
- Type d'attaches : Tie-on
-
Colors :
 Blue
Blue
- Size / Dimensions : 175 x 95 mm
- Option : Foam strip + Anti-glare strip + Visor
- Applications : Health, Medical
- Made in : France
Product information
Product features
| Article | Size | Color |
|---|---|---|
| M34101MNV-30 | 175 x 95 mm |  Blue Blue |
Packaging Features
| Inner case | Packaging |
|---|---|
| Dispenser box | 4 x 25 units |
Certifications & standards
- ISO 9001 Manufacturing site
- ISO 13485 Manufacturing site
- Clean room ISO 8 production
- Compliant to EU Regulation 2017/745 on Medical Devices
- ISO 10993-5
- ISO 10993-10
- Assessment of microbial contamination according to ISO 11737 : 2018 + A1 : 2021